Sustainable Polymers and Plastic Recycling
Our work in the area of sustainable polymers and plastic recycling is focussed on the development of ‘green’ strategies for the synthesis and functionalisation of polymers. These challenges present important targets from both a sustainability and biomaterials viewpoint. We have several strands to our research in this area that include derivation of new monomers that will result in materials with useful properties from renewable sources. As part of this work, we develop catalysts and efficient routes to their synthesis and devise efficient chemistries to modify them. The lack of existing materials and of commercially available monomers that enable the synthesis of functional degradable polymer backbones for advanced technological applications, as well as the need for sustainable polymer materials drives our interest in this area.
Plastics Recycling

What happens to plastics at the end of their useful life is a critical question that faces society. New approaches are required to both create new plastics with comparable or better properties to those that we currently use but that can feed into the circular economy but also to develop innovative strategies to recycle and upcycle existing plastic waste into high value products. Our primary approach is to consider discarded plastic (both in bulk as in the form of microplastics) as a raw material to produce high value molecules and materials to deliver new solutions to high value problems in energy (i.e. plastic electronics), pharmaceuticals (i.e. synthons for drugs), composites (i.e. lightweighting) and healthcare (i.e. biomaterials). See below for some of our key publications
Selective Chemical Upcycling of Mixed Plastics Guided by a Thermally Stable Organocatalyst. Jehanno, C.; Demarteau, J.; Mantione, D.; Arno, M. C.; Ruipérez, F.; Hedrick, J. L.; Dove, A. P.; Sardon, H. Angew. Chem. Int. Ed. (2021) 60, 6710 — 6717.
Synthesis of Functionalized Cyclic Carbonates through Commodity Polymer Upcycling. Jehanno, C.; Demarteau, J.; Mantione, D.; Arno, M. C.; Ruipérez, F.; Hedrick, J. L.; Dove, A. P.; Sardon, H. ACS Macro Lett. (2020) 9, 443 — 447.
Dual-catalytic depolymerization of polyethylene terephthalate (PET). Delle Chiaie, K. R.; McMahon, F. R.; Williams, E. J.; Price, M. J.; Dove, A. P. Polym. Chem. (2020) 11, 1450 — 1453.
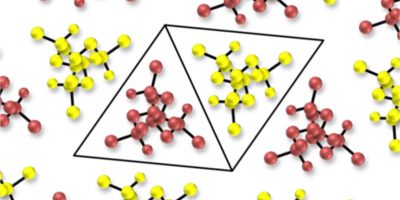
Polymerisation Catalysis
The group is focussed on the design, synthesis and study of the behaviour of both new organic and inorganic compounds that can be applied in polymerization catalysis. Our efforts in new catalyst design are focussed on molecules that can mediate both polymerization and depolymerisation and typically we are focussed on using step-growth or ring-opening polymerisation methodologies. Our interests specifically focus on the design of species that can mediate challenging transformations as well as provide high activity, target stereospecificity, and have high temperature stability and functional group tolerance. Overcoming such problems is a key challenge in the development of new materials from renewable sources as well as in depolymerisation of plastic waste. See below for some of our key publications.
Selective Organocatalytic Preparation of Trimethylene Carbonate from Oxetane and Carbon Dioxide. Huang, J.; Jehanno, C.; Worch, J. C.; Ruiperez, F.; Sardon, H.; Dove, A. P.; Coulembier, O. ACS Catal. (2020) 10, 5399 — 5404.
Organocatalysed depolymerisation of PET in a fully sustainable cycle using thermally stable protic ionic salt. Jehanno, C.; Flores, I.; Dove, A. P.; Muller, A. J.; Rupierez, F.; Sardon H. Green Chem. (2018) 20, 1205 — 1212.
Dual Catalysis for Selective Ring-Opening Polymerization of Lactones: Evolution toward Simplicity. Naumann, S.; Scholten, P. B. V.; Wilson, J. A.; Dove, A. P. J. Am. Chem. Soc. (2015) 137, 14439 — 14445.
Synthesis and Functionalisation of Sustainably-Sourced Polymers

Our primary foci are centred on both the development of novel degradable polymer materials from renewable/ sustainable resources such as malic acid, carbon dioxide, sugars and amino acids and also the application of highly efficient and orthogonal (‘click’) chemistries for the post-polymerization modification of degradable polymers. As part of this strategy, the incorporation of ‘click’-functional handles and the evaluation of post-polymerization functionalization strategies (without polymer degradation) means that a wider range of functional materials can be realized from relatively few sources. In turn, the functional groups will affect both the physical and degradation properties. See below for some of our key publications.
Modular Functionalization of Laminarin to Create Value-Added Naturally Derived Macromolecules. Costa, A. M. S; Rodrigues, J. M. M.; Perez-Madrigal, M. M.; Dove, A. P.; Mano, J. F. J. Am. Chem. Soc. (2020), 142, 19689 — 19697.
Selective Organocatalytic Preparation of Trimethylene Carbonate from Oxetane and Carbon Dioxide. Huang, J.; Jehanno, C.; Worch, J. C.; Ruiperez, F.; Sardon, H.; Dove, A. P.; Coulembier, O. ACS Catal. (2020) 10, 5399 — 5404.
Terpene- and terpenoid-based polymeric resins for stereolithography 3D printing. Weems, A. C.; Delle Chiaie, K. R.; Worch, J. C.; Stubbs, C. J.; Dove, A. P. Polym. Chem. (2019) 10, 5959 — 5966.