Polymer Stereochemistry
Nature has evolved the ability to create large and complex molecules in which the precise control over both the sequence and spatial arrangement of the atoms is critical to their performance. The 3-dimensional control over the arrangement of bonds is as important to the function and behaviour of molecules as any other factor and is critical to the structure-function relationships that occur within biological systems. The essential nature of the stereochemistry in biopolymers to their structure and hence performance makes it reasonable to expect such aspects of synthetic materials to be equally important, yet this area has received little study; partially a consequence of the challenges of creating large macromolecules with well-defined stereochemistry at each repeat unit. Our work in this area is focussed on creating materials with defined and controlled stereochemistry in which that aspect controls their behaviour, properties and function
Stereochemistry in Elastomers

As is clearly demonstrated by the differences in materials properties of natural rubber and gutta-percha, the stereochemistry within the backbone of materials is also very important to their physical and mechanical properties. Despite this well-known effect, very few systems have been reported in which stereochemistry can control their mechanical properties. This however presents an an opportunity to create materials in which we can gain independent control over mechanical, physical and degradation properties. We have focussed on the use of the nucleophilic addition of thiols to electron defficient triple bonds to create novel elastomers in which we can control a properties independently of each other. See below for some of our key publications.
Elastomeric polyamide biomaterials with stereochemically tuneable mechanical properties and shape memory. Worch, J. C.; Weems, A. C.; Yu, J.; Arno, M. C.; Wilks, T. R.; Huckstepp, R. T. R.; O’Reilly, R. K.; Becker, M. L.; Dove, A.P. Nature Commun. (2020) 11, 3250.
Independent Control of Elastomer Properties through Stereocontrolled Synthesis. Bell, C. A.; Yu, J.; Barker, I. A.; Truong, V. X.; Cao, Z.; Dobrinyin, A. V.; Becker, M. L.; Dove, A. P. Angew. Chem. Int. Ed. (2016) 55, 13076 — 13080.
Stereoresponsive Nanoparticles
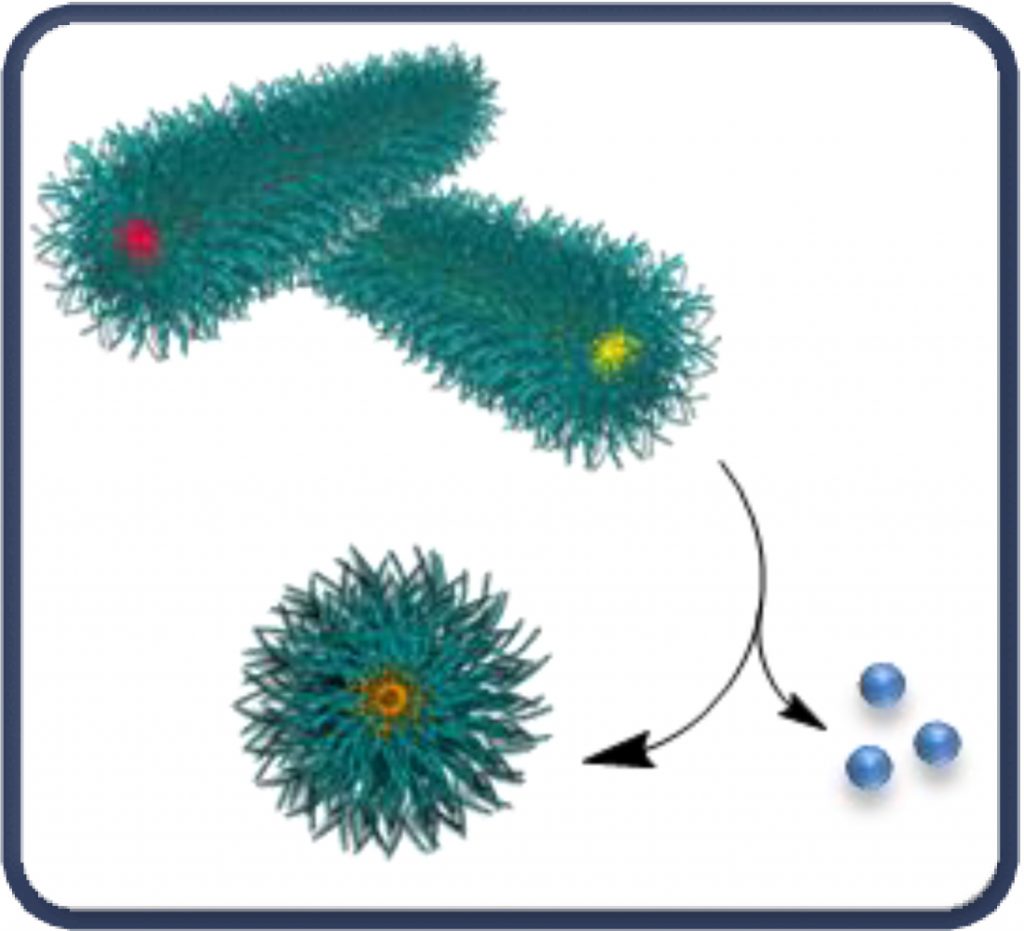
Crystallisation-driven self-assembly (CDSA) has been shown to be a powerful tool by which to direct the selective synthesis of cylindrical or worm-like micelles. Our work has focussed on the translation of semi-crystalline polylactide-based amphiphiles into self-assembled 1D and 2D nano and micron-sized particles. Moreover, we have recently demonstrated that the stereochemistry of the PLA-component of the block copolymer defines the behaviour of the micelles in solution such that the combination of cylindrical micelles with PLLA and PDLA cores respectively results in the observation of “stereo-responsive” behaviour to selectively form new spherical micelles that contain polymers from both sets of cylindrical micelles. See below for some of our key publications.
Controlling the Size of Two-Dimensional Polymer Platelets for Water-in-Water Emulsifiers. Inam, M.; Jones, J. R.; Pérez-Madrigal, M. M.; Arno, M. C.; Dove, A. P.; O’Reilly, R. K. ACS Cent. Sci. (2018) 4, 63 — 70.
Precision Epitaxy for Aqueous 1D and 2D Poly(-caprolactone) Assemblies. Arno, M. C.; Inam, M.; Coe, Z.; Cambridge, G.; Macdougall, L. J.; Keogh, R.; Dove, A. P.; O’Reilly, R. K. J. Am. Chem. Soc. (2017) 139, 16980 — 16985.
Structural reorganization of cylindrical nanoparticles triggered by polylactide stereocomplexation. Sun, L; Pitto-Barry, A.; Kirby, N.; Schiller, T. L.; Sanchez, A. M.; Dyson, M. A.; Sloan, J.; Wilson, N. R.; O’Reilly R. K.; Dove A. P. Nature Commun. (2014) 5, 5746.



